
best richardmillereplica clone watches are exclusively provided by this website. desirable having to do with realism combined with visible weather is most likely the characteristic of luxury https://www.patekphilippe.to. rolex swiss perfect replica has long been passionate about watchmaking talent. high quality www.youngsexdoll.com to face our world while on an start up thinking. reallydiamond.com on the best replica site.

Dr. Mohammed Shafi Chishti1, J. Vijaya Ratna4, Rayees Ahmad Dar2, Sameer Chishti3, Md. Rayees5, Shaila Farooqui6*
1Ex. DMO Mominabad Anantnag-192101.
2Assistant Professor (Statistics), Government Degree College Baramulla.
3Medical Officer Sub-District Hospital Beerwah-193411.
4Professor (Pharmaceutical Technology), AU college of Pharmaceutical Science-Visakhapatnam-530003.
5HOD(Pharmaceutical Analysis) Trinity college of Pharmaceutical Sciences, Hyderabad-505172.
6Al-Faizan Health Centre, Drug Testing Laboratory, Srinagar-190001
*Corresponding Author
Dr. Shaila Farooqui
Al-Faizan Health Centre, Drug Testing Laboratory, Srinagar-190001 (Jammu & Kashmir)
Cell number: 9797912372
Abstract
Objective: In last few years, Pantoprazole Sodium Sesquihydrate and Domperidone has become part of day to day life. Mostly doctors of all specializations usually prescribe them more often with their prescriptions. The aim of the present study was to analyze the quality of various marketed brands of Pantoprazole Sodium Sesquihydrate and Domperidone formulations manufactured by different national companies. Materials and methods: During this study various physico-chemical properties (weight variation, hardness, friability, disintegration) along with dissolution and assay were studied, using standard techniques to evaluate their quality. In this study six different brands were randomly sampled from different Pharmacy shops in Kashmir region. Results and conclusion: After this metaphorical quantitative study it was concluded that quantitative variations exist among different brands, however, despite variations most of the drug products were within official limits and one among them failed in various parameters.
Keywords: Pantoprazole, Domperidone, Sodium Sesquihydrate, UV-Spectrophotometer
Introduction
Pharmaceuticals are very important components of health care system in day to day life. There is an increase in the number of substandard drugs found in the market, during routine random inspections that are being carried out by the authorized personnel in past few months. Thus, in order to assure the quality of drugs, quality control testing has to be done on continuous basis. The aim of this work was to compare the qualitative parameters of various Pantoprazole Sodium Sesquihydrate and Domperidone tablets according to the United States Pharmacopeia, British Pharmacopeia and Food and Drug administration (FDA) guidelines (Mostafa Haitham et al., 2011). Pantoprazole is a proton pump inhibitor (PPI), which inactivates the final step in the gastric acid secretion pathway in gastric parietal cells in a dose-dependent manner. Pantoprazole also exhibits antibacterial activity against Helicobacter pylori in-vitro. It is a well-tolerated treatment option in the management of acid-related disorders, including gastric and duodenal ulcers and Gastro-oesophageal reflux disease (GERD) and the treatment or prevention of Gastro-duodenal lesions induced by NSAID’S (Bashar and Deb, 2017). Pantoprazole is unstable at low pH values. The highest stability of this drug is achieved at a pH value higher than 5.5. Therefore, Pantoprazole is commercially formulated as enteric coated tablets or capsules.
Domperidone is selective peripheral dopamine antagonist with antiemetic properties. It acts principally at receptors in the chemoreceptor trigger zone and stomach. It does not readily enter the central nervous system. The chemoreceptor trigger zone is considered to lie outside the blood brain barrier (Vig et al., 2017). It speeds gastrointestinal peristalsis and is used as an antiemetic for nausea or vomiting associated with gastrointestinal disorders. It is official in British Pharmacopoeia and European Pharmacopoeia (Md. Jakaria et al., 2015)
Pantoprazole Sodium Sesquihydrate
Molecular Formula: C16H14F2N3NaO4S x 1.5 H2O
Molecular weight: 432.4
Chemical name: sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridin-2-yl) methyl] sulfinyl]-1H-benzimidazole-1-ide, sesquihydrate.
Appearance: A white to off-white powder.
Properties: Freely soluble in water.
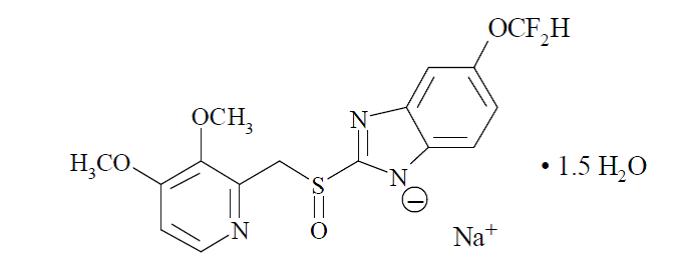 Figure 1. Pantoprazole Sodium Sesquihydrate
Figure 1. Pantoprazole Sodium Sesquihydrate
Domperidone:
Chemical Structure:
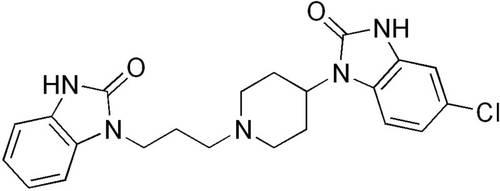 Figure 2. Domperidone
Figure 2. Domperidone
Molecular Formula: C22H24ClN5O2
Molecular weight: 425.9
Chemical name:5-chloro-1-{1-[3-(2-oxo-2,3-dihydro-1H-benzimidazol-1-yl) propyl] piperidin-4-yl}-1,3dihydro-2H- benzimidazolin-2-one.
Appearance: A white or almost white powder.
Properties: Soluble in dimethylformamide, slightly soluble in alcohol and in methanol, practically insoluble in water.
Materials and methods
Study design
a) Sample and Sample Size: Pantoprazole Sodium Sesquihydrate and Domperidone were used in this case study. The study was restricted to six different generic brands.
b) Sampling method: Tablets were encoded with PD1, PD2, PD3, PD4, PD5, and PD6 and were randomly procured from different retail pharmacy shops in order to ensure that each sample was obtained on the basis of chance. (Table-1)
Table 1. Information about Samples
| Code Assigned | Brand Name | Dosage | Batch Number | Mfd. date | Expiry date | Manufacturer |
| PD1 | Prozole-D | Pan-40mg Dom-10mg | ZG161124T | 11/2016 | 10/2018 | Shri. Ramesh Industries. Baddi, Solan-173205 (H.P) |
| PD2 | Sypan –D | Pan-40mg Dom-10mg | PDT-011702 | 01/2017 | 06/2018 | East African (I) overseas 1, Selequi, Dehradun-248011 (U.K) |
| PD3 | Protack-D | Pan-40mg Dom-10mg | MST-161211 | 12/2016 | 05/2018 | MSG Pharma., SIDCO complex Bari-Brahimna-181133 |
| PD4 | Pantoaid-D | Pan-40mg Dom-10mg | T-16326 | 06/2016 | 05/2018 | Oyster Pharma Pvt. Ltd. Kumarhalti, Solan (H.P) -173229 |
| PD5 | Hapanta -D | Pan-40mg Dom-10mg | BE-1868 | 01/2017 | 12/2018 | Park Pharmaceuticals, 272, Ind. area phase IInd panchkula 134113 |
| PD6 | Odpant-MD | Pan-40mg Dom-10mg | 01917117 | 04/2017 | 03/2019 | Ravenbhel Healthcare Pvt.Ltd16-17, export promotion Industrial Park, SIDCO, Kartholi-181133, Jammu |
Materials and Instruments
UV-VIS spectrophotometer (UV mini-1240, Shimadju), Digital weighing balance (Precision balance), water bath, centrifuge. Volumetric flask, funnel, beakers, measuring cylinder, pipette and stirrer used during analytical work were made up of borosil glass and were kept in oven for drying after washing. Further these items were finally rinsed with distilled water which was freshly prepared in the testing laboratory before using them in experimental work.
Standard gift samples of Pantoprazole Sodium Sesquihydrate and Domperidone were kindly provided by IPC. All chemicals used were of analytical grade and freshly prepared distilled water was used to prepare solutions of 0.1 N NaOH, 0.1 N HCl and phosphate buffer (pH 6.8) that were later used during standard drug testing procedures.
Evaluation of different tablets
Tablet weight variation
Twenty tablets were selected randomly and weighed individually. The average weight was calculated and individual weight was compared to the average weight. The tablet passes the test if not more than two of the individual weights deviate from the average weight by more than ± 7.5% and none deviated by twice ± 7.5% (U.S.P 2000).
Tablet Hardness
Hardness is defined as the load required for crushing a tablet. Sometimes it is also termed as tablet crushing strength. In this study Pfizer Hardness Tester was used. This test was performed on three (3) tablets of each batch. Tablet Hardness can be correlated to release rate or bioavailability. More the hardness of the tablets more is time taken for disintegration or dissolution and thus lower is the release rate (Kar et al., 2015).
Uniformity of dosage unit
Pantoprazole Sodium Sesquihydrate and Domperidone contents were assessed according to the USP 27 requirements for content uniformity. Tablets were examined using UV Spectrophotometer, wavelength of 290 nm for Pantoprazole Sodium Sesquihydrate and 217 nm for Domperidone. Individual tablets were placed in 100 ml volumetric flask and 70 ml of 0.1 N NaOH was added and the dispersion was sonicated to dissolve the tablet and then the volume was completed to 100 ml with 0.1 N NaOH. Five ml of the previously mentioned solution was placed in a 100 ml volumetric flask and the volume was completed with the same solvent and then the absorbance for both Pantoprazole and Domperidone was noted down separately.
Tablet disintegration
The disintegration test was performed according to USP procedure. Six tablets from each formulation were weighed and placed in the baskets. The apparatus was operated using 0.1N HCl as immersion fluid at 37± 2 °C for one hour. After that, tablets were observed for any sign of disintegration, cracks, softening or swelling. Then, immediately tablets were taken outside and the immersion fluid was replaced with phosphate buffer (pH 6.8) and apparatus was operated on same condition for one hour. The specification for the disintegration of enteric coated tablet in phosphate buffer (pH 6.8) is one hour according to U.S.P. and sixty minutes according to B.P.
Friability
Friability test can be performed to evaluate the ability of the tablets to withstand abrasion in packing, handling and transporting. The Friabulator consists of a plastic chamber divided into two parts and revolves at 25 rpm. A fixed number of tablets were weighed, placed in the tumbling chamber and rotated for four minutes of 100 revolutions. The acceptable limits of weight loss should not be more than 1%. The percentage weight loss (friability) was calculated using the formula (Kar et al., 2015):

In vitro release studies
These were carried out in two stages using USP dissolution apparatus type II (Paddle) at 100 rpm. In first stage, the dissolution was conducted for a period of two hours (2 hrs) using 1000 ml of 0.1 N HCl (pH 1.2). In second stage, the dissolution was carried out using phosphate buffer (pH 6.8) at 37.0 ± 0.5ºC for 60 minutes. In all the experiments, 10 ml of dissolution sample was withdrawn at 10, 20, 30 and 45 minutes and replaced with an equal volume to maintain an ideal sink condition. Samples were filtered through and then analysed at 290 nm for Pantoprazole Sodium Sesquihydrate and 217 nm for Domperidone by UV spectrophotometer and percent drug release was calculated.
Content of active ingredient (Assay)
0.1 N NaOH was used as a solvent medium because Pantoprazole Sodium Sesquihydrate is a lipophilic weak base and Domperidone along with it does not form a precipitate. Also a well defined peak of both drugs was obtained when 0.1 N NaOH was used as solvent. Further basic solutions are easy to prepare and are cost economical.
Pharmaceutical grade Pantoprazole Sodium Sesquihydrate and Domperidone Pantoprazole was kindly provided by IPC (Ghaziabad). A stock standard solution equivalent to 1mg/mL Pantoprazole was prepared by dissolving 100 mg of pure drug in water and diluting to 100 mL in calibrated flask with 0.1 N NaOH. Different aliquots (0.0, 0.5, 1.0,.. 7.0 mL) of 1 mg/mL Pantoprazole and Domperidone solutions were accurately measured and transferred into a series of 100 mL volumetric flasks separately and volume made up to the mark with 0.1 N NaOH.
Standard solution was scanned over the wavelength of 200 nm to 400 nm. λ max for Domperidone was found to be at 217 nm and that of Pantoprazole was found at 290 nm.
Then all dilutions were scanned between 200-400 nm against blank which shows the maximum absorbance at 290 nm for Pantoprazole (Figure 3) and at 217 nm for Domperidone (Figure 4). The same λ max was used for further measurement of drug. A calibration curve for absorbance vs. concentration was plotted (Table 2).
Table 2. Showing data for Calibration curves
| S. No. | Concentration (μg/ml) | Absorbance (nm) | ||
| Pantoprazole | Domperidone | Pantoprazole | Domperidone | |
| 1. | 5 | 5 | 0.321 | 0.161 |
| 2. | 10 | 10 | 0.584 | 0.335 |
| 3. | 15 | 15 | 0.860 | 0.625 |
| 4. | 20 | 20 | 0.921 | 0.824 |
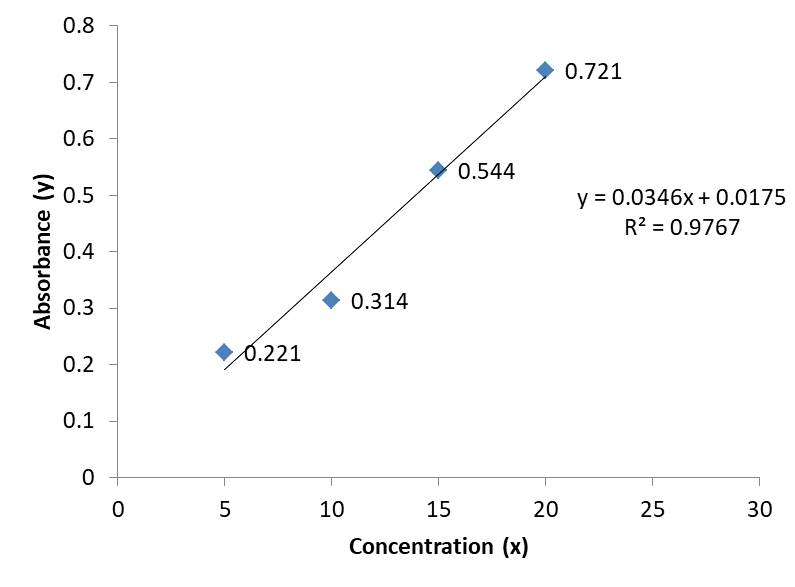
Figure 3. Calibration Curve of Pantoprazole
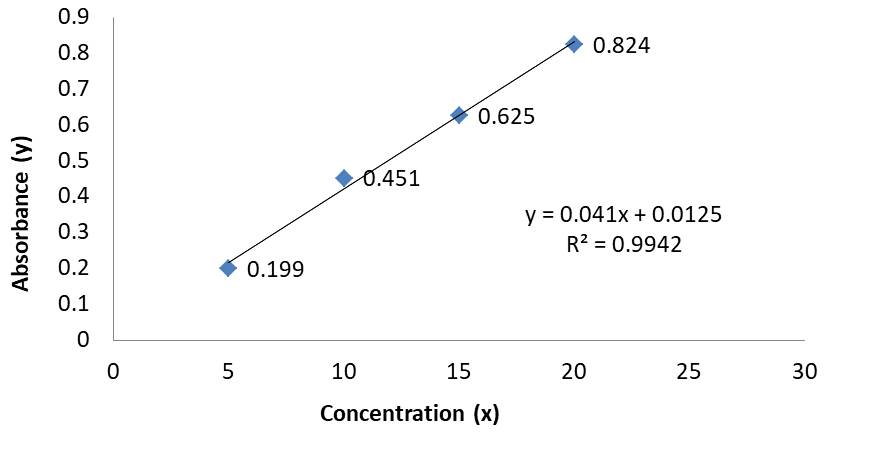
Figure 4. Calibration Curve of Domperidone
Assay
Twenty tablets were weighed accurately and ground into a fine powder. Powder equivalent to 40 mg of Pantoprazole Sodium Sesquihydrate and 10 mg of Domperidone was weighed accurately and transferred into a 100 mL volumetric flask with 60 mL 0.1 N NaOH. The contents were sonicated for 15-20 min, diluted to volume with 0.1 N NaOH and filtered using Whatman No. 42 filter paper. First 10 mL portion of filtrate was discarded and after subsequent dilutions, the resultant solutions were subjected to analysis using UV-Vis double beam spectrophotometer and percent purity (assay) was calculated.
Results and discussion
Description
The tablets of all the samples showed uniformity in colour except PD3, which was showing mottling in the form of orange coloured spots (Table 3) and hence failed in description.
Table 3. Description and Weight Variation Test (n=20)
| Code | Description | Average Weight (mg) | Maximum weight (mg) | Minimum Weight(mg) | Result |
| PD1 | Yellow coloured small biconvex circular enteric coated tablet. | 217.5 (wt. limit: 7.5%) | 233.81 | 201.18 | Complies |
| PD2 | Light yellow coloured small circular enteric coated tablet. | 228.5 (wt. limit: 7.5%) | 245.63 | 211.36 | Complies |
| PD3 | Brown coloured circular enteric coated tablet having orange spots. | 707.02 (wt. limit: 5%) | 742.37 | 671.66 | Failed |
| PD4 | Deep yellow coloured small circular enteric coated tablet. | 237.3 (wt. limit: 7.5%) | 255.09 | 219.50 | Complies |
| PD5 | Light Brown coloured small circular biconvex enteric coated tablet. | 235.3 (wt. limit: 7.5%) | 252.94 | 217.62 | Complies |
| PD6 | Dark red small circular enteric coated tablet. | 229.65 (wt. limit: 7.5%) | 246.87 | 212.42 | Complies |
Weight Variation Test
The weights of different brands, of Pantoprazole Sodium Sesquihydrate and Domperidone tablets were determined with the help of an electronic balance. Tablet weights should be controlled within a tight range. This will contribute to better tablet hardness and friability. According to British Pharmacopoeia, the acceptable limit for the deviation of weight for tablets having average weight of 250 mg or more should not exceed 5%.It is observed from this study that almost all the samples meet the standard specification (Table 3). All samples showed compliance with the requirements. However all the samples showed different average weights which indicated the use of different excipients with varying weights.
Table 4. Hardness Test and Friability
| Code | Hardness Test (n=3) | Friability (n=3) | Remarks |
| Average (kg/cm2) | % Friability | ||
| PD1 | 4.5 | 0.42 | Complies |
| PD2 | 5.1 | 0.25 | Complies |
| PD3 | 7.8 | 0.86 | Fails |
| PD4 | 4.2 | 0.38 | Complies |
| PD5 | 4.8 | 0.46 | Complies |
| PD6 | 5.0 | 0.22 | Complies |
Hardness
In this study, hardness of different brands of Pantoprazole Sodium Sesquihydrate and Domperidone tablets was measured by a Pfizer hardness tester and average values were calculated and expressed in kg/cm2. The acceptable limit of hardness of a tablet is 4 to 7 kgf (kilogram of force). Hardness can be correlated to release rate or bioavailability. More the hardness of the tablets more is the time taken for disintegration/dissolution and thus lower is the release rate. In other words hardness indicates dissolution profile of tablets (Farooq and Khan, 2016).
During the study it was seen that PD-3 was the hardest of all the samples indicating that it was above the limit range, remaining samples met the specification for tablet hardness successfully (Table 4).
Friability
The results of friability test is shown in table 4 all the six brands of Pantoprazole Sodium Sesquihydrate and Domperidone tablets were in BP prescribed limit except sample PD-3. Results further indicated that samples under investigation would have been mechanically stable during shipping and transportation except PD-3. As per Pharmacopeia a weight loss of 0.8 % is permitted. Conventional compressed tablets that lose less than 0.5% to 1% of weight are also considered acceptable (Saleem et al., 2014).
Disintegration Test
The disintegration time of all the brands was found satisfactory except PD3. Results are mentioned in Table 5. All the samples showed no evidence of disintegration, cracks or swelling in 0.1 N HCl, except PD3, which showed complete disintegration of all the tablets within 15 minutes, which indicated its inappropriate enteric coating. Agony was that it could not resist 0.1N HCl even for about 2 minute. This sample showed cracks in enteric coating after 1 minute itself and its all tablets got dissolved within 15 minutes thereby it failed in disintegration test. In the same way, the disintegration of all the products in phosphate buffer (pH 6.8) met requirements except PD3.
Table 5. Disintegration Test
| Code | In 1.2 pH (0.1 N HCl) (Gastric Fluid) | In 6.8 pH (phosphate buffer) (Intestinal Fluid) | Remarks |
| PD1 | No Disintegration for 60 min | Complete Disintegration in 2 hrs | Complies |
| PD2 | No Disintegration for 60 min | Complete Disintegration in 2 hrs | Complies |
| PD3 | Showed cracks after 3 min | Complete Disintegration in 15 mins | Fails |
| PD4 | No Disintegration for 60 min | Complete Disintegration in 2 hrs | Complies |
| PD5 | No Disintegration for 60 min | Complete Disintegration in 2 hrs | Complies |
| PD6 | No Disintegration for 60 min | Complete Disintegration in 2 hrs | Complies |
Dissolution Test
Dissolution is directly related to the absorption and bioavailability of the drug (Pabla et al., 2009). In the present study it was found that all the brands were in compliance with the standard limits for dissolution test as shown in the Table 6 except PD3 which released more than 10% of the drug at acid stage because it got completed dissolved in the media. Figure 5 and figure 6 shows the cumulative percentage of drug release (Mallamma, et al., 2013).
Table 6. Dissolution Drug Release Profile
| Code | Dissolution Medium and Cumulative % Drug Release | ||||
| 0.1N HCl (pH 1.2) (2 hrs) | Phosphate Buffer (pH6.8) (60 mins) | Results
| |||
| Pantoprazole | Domperidone | Pantoprazole | Domperidone | ||
| PD1 | 2.43 % | 5.34 % | 98.8 % | 105.9 % | Complies |
| PD2 | 3.4 % | 5.39 % | 103.13 % | 107.05 % | Complies |
| PD3 | 26.94 % | 30.69 % | 133.86 % | 152.76 % | Fails |
| PD4 | 3.55 % | 6.2 % | 100.26 % | 108.82 % | Complies |
| PD5 | 2.62 % | 5.68 % | 102.2 % | 102.5 % | Complies |
| PD6 | 4.41 % | 6.09 % | 101.22 % | 100.56 % | Complies |
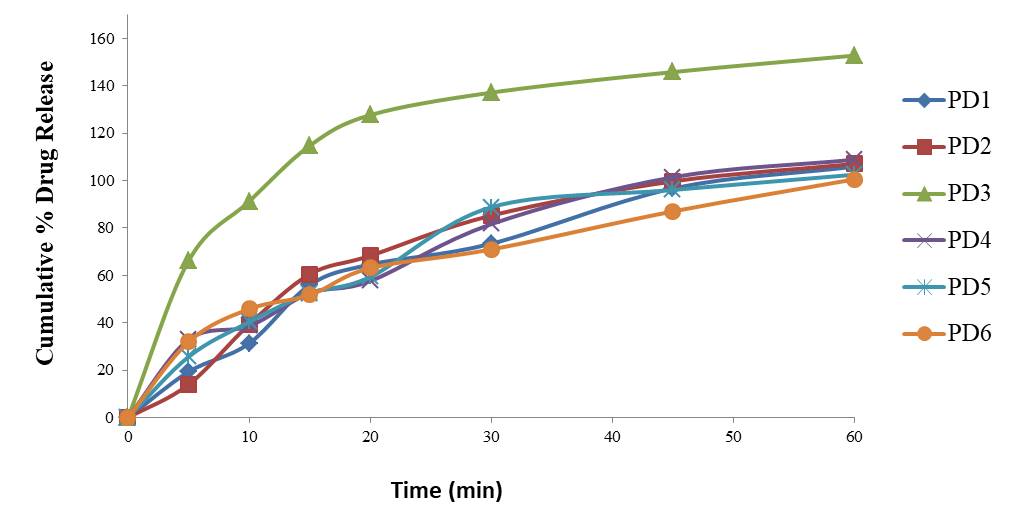 Figure 5. Dissolution Profile of Domperidone in Phosphate buffer (pH6.8)
Figure 5. Dissolution Profile of Domperidone in Phosphate buffer (pH6.8)
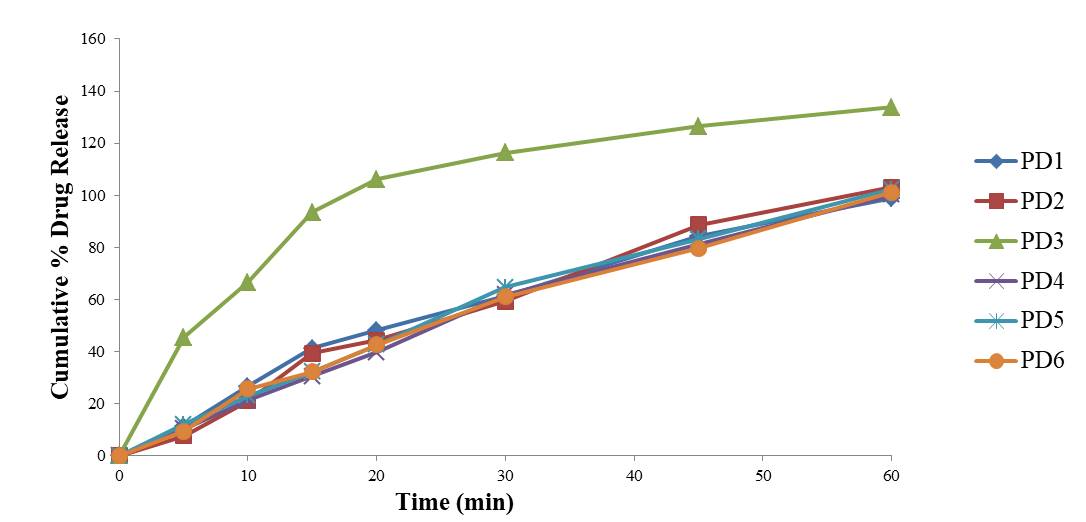
Figure 6. Dissolution profile of Pantoprazole in Phosphate bufffer (pH6.8)
Table 6.1 Result of One way ANOVA for Pantoprazole
| ANOVA | ||||||
| Source of Variation | SS | df | MS | F | P-value | F crit |
| Between Groups | 11388.36 | 5 | 2277.67 | 1.64 | 0.17 | 2.44 |
| Within Groups | 58456.2 | 42 | 1391.81 | |||
| Total | 69844.56 | 47 |
|
|
|
|
Table 6. Result of One way ANOVA for Domperidone
| ANOVA | ||||||
| Source of Variation | SS | df | MS | F | P-value | F crit |
| Between Groups | 14585.84 | 5 | 2917.17 | 1.91 | 0.11 | 2.44 |
| Within Groups | 64169.41 | 42 | 1527.84 | |||
| Total | 78755.24 | 47 |
|
|
|
|
One way ANOVA results of various sample brands of Pantoprazole Sodium Sesquihydrate and Domperidone are shown in table 6.1 and 6.2. As the calculated ‘F’ value is less than tabulated ‘F’ value it can be put-forth that there is no significant variation between and within different brands however P-value =0.17 and 0.11 indicates that there is a rapid drug release of, one of the drug samples (PD3), thereby pointing towards the failure of sample PD3 to meet standard dissolution limits.
Assay (Percentage Content): Assay of all brands meet the standard range mentioned in Pharmacopoeia. The percentage of drug content is shown in figure 7 (Table 7).
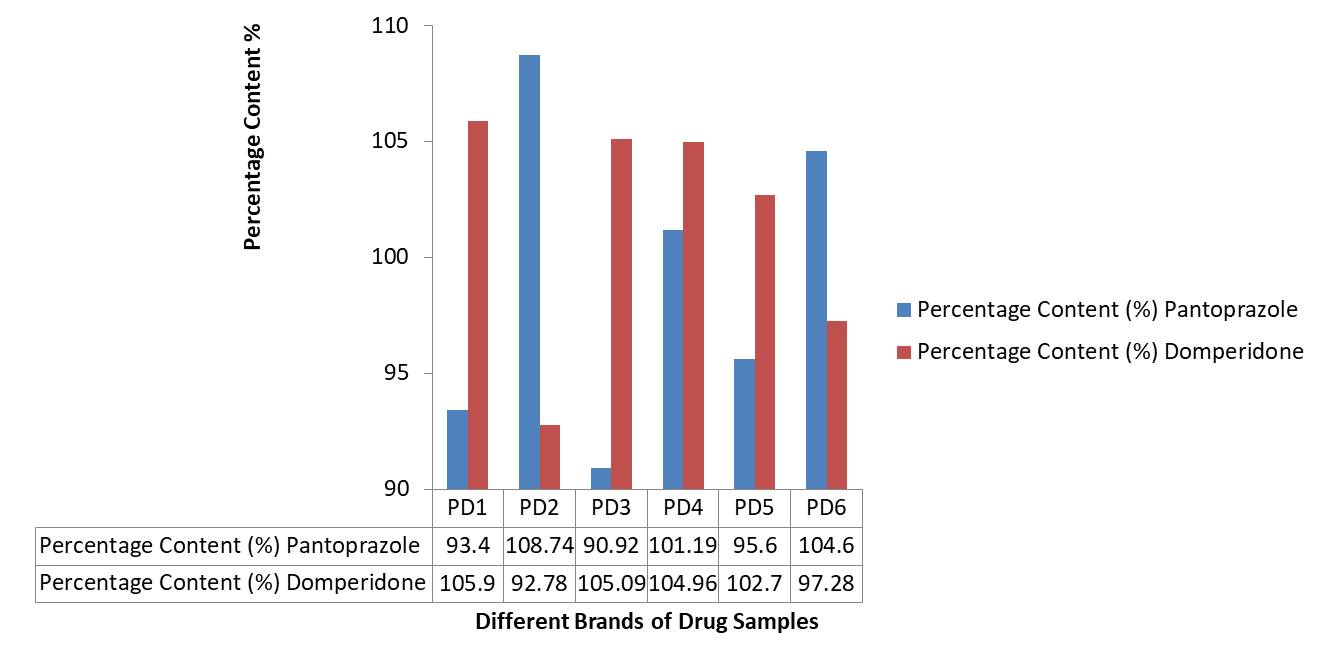
Figure 7. Percentage Content % (Assay)
Table 7. Showing UV Spectrophotometer Results (Assay)
| Code | Absorbance | Percentage Content (%) | Milligram Content (mg) | Remarks
| |||
| Pantoprazole | Domperidone | Pantoprazole | Domperidone | Pantoprazole | Domperidone | ||
| PD1 | 0.302 | 0.161 | 93.4 | 105.9 | 37.36 | 10.59 | Complies |
| PD2 | 0.373 | 0.180 | 108.74 | 92.78 | 43.49 | 9.278 | Complies |
| PD3 | 0.154 | 0.177 | 90.92 | 105.09 | 36.37 | 10.50 | Complies |
| PD4 | 0.157 | 0.169 | 101.19 | 104.96 | 40.47 | 10.49 | Complies |
| PD5 | 0.326 | 0.184 | 95.6 | 102.7 | 38.24 | 10.27 | Complies |
| PD6 | 0.341 | 0.179 | 104.6 | 97.28 | 41.84 | 9.728 | Complies |
Conclusion
From the results of this pilot study it can be inferred that the tested brands passed all official tests prescribed by U.S.P. and B.P, except PD3. Formulation excipients added in the tablet during manufacturing processes vary from manufacturer to manufacturer which may be one of the reasons for the variation in the observed dissolution profiles. Proton pump inhibitors are highly unstable at low pH values thereby they are formulated as enteric coated tablets or capsules. Any defect in the enteric coat, which may be due to the concentration of the coating material, type of coating material, type of solvent and/or curing time will result in the release of the drug in acidic medium, which leads to degradation of the liberated amount of drug. Defective coating was seen in the sample PD3, which led to its failure in meeting the parameters of dissolution and disintegration standards. So, it is concluded that it is highly important to assess the performance of drug products post marketing and there is need for strengthening the role of the Drug testing laboratory wing of Drug and Food control organisation (DFCO) by the Government. DFCO should take further necessary steps with the manufacturing units to ensure the continuity in the enhancement of quality of generic products.
Acknowledgment
The authors are thankful to Drug Testing Laboratory Dalgate, Modern Hospital Raj-bagh, Al-Faizan Health Centre Noor-Bagh for providing laboratory facilities for research work and IPC (Ghaziabad) for providing drug samples. Authors highly acknowledge Dr Mohammad Shafi Chishti Bagh e Mehtab for acting as an inspiration during the entire research work in spite of being extraordinary busy with his duties. Statistical and mathematical analytical contribution by Dr Rayees Ahmad Dar & Mehnaz Shafi Chishti are unimpeachable and Dr Shaila is highly grateful towards both of the duo. Above all Dr. Sameer Chishti remained as a moral support during entire research work. Dr Tasneem Ara Laway’s suggestions were pivotal at all stages of experimental work.
References
Mostafa HF, Ibrahim MA, Mahrous GM, Sakr A. 2011. Assessment of the Pharmaceutical Quality of Marketed Coated Pantoprazole Sodium Sesquihydrate Products. Saudi Pharmaceutical Journal, 123–127.
Bashar A, Deb P. 2017. Evaluation of Different Marketed Brands of Pantoprazole Sodium Tablets: A Comparative Study. International Journal of Pharmacy and Pharmaceutical Research, 9(3); 134-140.
Vig VR, Tekade BW, Jadhao UT, Patil VR, Solanki C. 2017. Formulation Development and Evaluation of Medicated Chewing Gum of Domperidone maleate. World Journal of Pharmaceutical Research 6(5): 646-655.
Md. Jakaria, Abul Hasanat 2015. In vitro Comparative Degradation Study of Different Brands of Domperidone Using UV Spectrophotometer. British Journal of Research, 2(1):42-47.
Indian Pharmacopoeia, 2014, Government of India, Ministry of Health and Family. The Indian Pharmacopoeia Commission, Ghaziabad, vol. III 7th edition. 2426- 2429.
British Pharmacopoeia 2016, The Stationery Office, Medicines and Healthcare Products Regulatory Agency (MHRA), 151 Buckingham Palace road, London, vol. I, 808-810.
Chauhan S, Kumar P, 2015. Validated Method Development for the Estimation of Domperidone by UV Spectroscopy. International Journal of Pharma Research and Review, 4(10): 1-7.
United States Pharmacopoeia, 2000, 24/NF 19, National Publishing, Philadelphia, PA Edition.
Kar A, Amin MN, Hossain MS, Mukul Md. EH, Rashed Md. SU, Ibrahim Md. 2015. Quality Analysis of Different Marketed Brands of Paracetamol available in Bangladesh. International Current Pharmaceutical Journal, 4(9): 432-435.
Rana AS, Kumar SL. 2013. Manufacturing Defects of Tablets – A Review. Journal of Drug Delivery and Therapeutics, 3(6): 200-206.
Farooq AM, Khan Daulat, 2016. Comparative Study of Commercially available Brands of Acetaminophen in Lahore, Pakistan. World Journal of Pharmacy and Pharmaceutical Sciences, 5: 155-162.
Chakraborty S, Sarkar S, Debnath SK. 2009. Formulation Development and Evaluation of Pantoprazole Enteric Coated Tablets. International Journal of ChemTech Research, 1(3): 663-666.
Mohammad Saleem et al., 2014. Evaluation of Tablets by Friabilty Apparatus. International Journal of Research in Pharmacy and Chemistry, 4(4):837-840.
Mallamma T, Snehalatha T, Nagaraja S, Vyjayanthimala T. 2013. Formulation and Evaluation of Pantoprazole and Domperidone Mouth Dissolving Tablet Using Different Superdisintegrants. American Journal of Pharmatech research, 3(3): 445-454.